Capreomycin, a polypeptide antibiotic complex of 4 microbiologically active components, is an antituberculosis agent. Capreomycin is used in conjunction with other antituberculosis agents in the treatment of clinical tuberculosis.
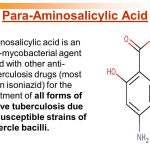
Aminosalicylic acid is used in conjunction with other antituberculosis agents in the treatment of clinical tuberculosis. The drug is designated an orphan drug by the US Food and Drug Administration (FDA) for use in the treatment of tuberculosis. These regimens have a minimum duration of 6 months (26 weeks), and consist of an initial intensive phase (2 months) and a continuation phase (usually either 4 or 7 months). Aminosalicylic acid is considered a second-line antituberculosis agent for use in these regimens.

Antituberculosis agents are antibiotics and synthetic anti-infectives used in the treatment of tuberculosis and other diseases caused by organisms of the genus Mycobacterium. Isoniazid, rifampin, ethambutol, and pyrazinamide are the drugs used most frequently in the treatment of tuberculosis and are considered first-line agents for use in antituberculosis regimens.

While flucytosine may be effective when used alone for the treatment of some fungal infections (e.g., candidal cystitis, chromomycosis), the drug should not be used alone in the treatment of severe, life-threatening infections. In the treatment of serious candidal or cryptococcal infections, flucytosine generally has been used as an adjunct to IV amphotericin B or an azole antifungal agent (e.g., fluconazole, itraconazole). There is some evidence that combined use of flucytosine and amphotericin B may be associated with an increased risk of serious adverse effects, especially in immunocompromised patients including those with human immunodeficiency virus (HIV) infection.
Nystatin is used orally for the treatment of intestinal candidiasis. In patients with coexisting intestinal candidiasis and vulvovaginal candidiasis, nystatin has been administered orally in conjunction with intravaginal application of an antifungal agent. Nystatin has been administered orally as a suspension in conjunction with local application of the drug for the treatment of candidal diaper dermatitis. The majority of infants with candidal diaper dermatitis harbor C. albicans in their intestines, and infected feces appear to be an important source of the cutaneous infection.

Systematic drug interaction studies have not been performed to date using amphotericin B cholesteryl sulfate complex, amphotericin B lipid complex, or amphotericin B liposomal. The fact that drug interactions reported with conventional IV amphotericin B could also occur with these lipid-based or liposomal formulations of the drug should be considered.

Acute infusion reactions (e.g., fever, chills, headache, nausea, vomiting) and nephrotoxicity are the most frequent adverse reactions to conventional IV amphotericin B. Although clinical experience with amphotericin B cholesteryl sulfate complex, amphotericin B lipid complex, and amphotericin B liposomal is limited to date, these drugs appear to be better tolerated than conventional IV amphotericin B.
Conventional amphotericin B is administered by IV infusion. The drug also has been given intra-articularly, intrapleurally, intrathecally, or by local instillation or irrigation. For information regarding administration of the drug as an oral suspension and administration of the drug topically, subconjunctivally, or by local instillation or irrigation.

Conventional IV amphotericin B (formulated with sodium desoxycholate) is used for the treatment of potentially life-threatening fungal infections including aspergillosis, North American blastomycosis, systemic candidiasis, coccidioidomycosis, cryptococcosis, histoplasmosis, paracoccidioidomycosis,, sporotrichosis, and zygomycosis.
Caspofungin acetate, a semisynthetic lipopeptide synthesized from a fermentation product of Glarea lozoyensis, is an echinocandin antifungal agent. The drug is a glucan synthesis inhibitor and differs structurally and pharmacologically from other currently available antifungal agents. Caspofungin inhibits the synthesis of b(1,3)-d-glucan, an integral component of the fungal cell wall that is not present in mammalian cells.