
Terbinafine hydrochloride is a synthetic allylamine antifungal agent. Terbinafine is structurally and pharmacologically related to naftifine.
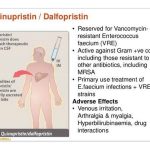
Quinupristin and dalfopristin is administered by IV infusion over 60 minutes. Quinupristin and dalfopristin powder for injection must be reconstituted and diluted prior to administration. The manufacturer states that only 5% dextrose injection or sterile water for injection should be used to reconstitute the powder, and further dilution should be with 5% dextrose. Strict aseptic technique must be observed since the drug contains no preservative.
The drug also is active in vitro against Arcanobacterium haemolyticum (formerly Corynebacterium haemolyticum). Clindamycin is active against some anaerobic and microaerophilic gram-negative and gram-positive organisms including Actinomyces, Bacteroides, Eubacterium, Fusobacterium, Propionibacterium, microaerophilic streptococci, Peptococcus, Peptostreptococcus, and Veillonella. Clindamycin is active in vitro against Prevotella and Porphyromonas (both formerly classified as Bacteroides); Mobiluncus (motile, anaerobic, curved rods) also are inhibited in vitro by the drug. Clostridium perfringens, C. tetani, Corynebacterium diphtheriae, and Mycoplasma are also inhibited by clindamycin.

Ototoxicity and nephrotoxicity are the most serious adverse effects of parenteral vancomycin therapy. To date, these adverse effects have not been reported in patients receiving vancomycin orally. The incidences of ototoxicity and nephrotoxicity have not been well established, but clinical experience to date suggests that these adverse effects occur relatively infrequently.